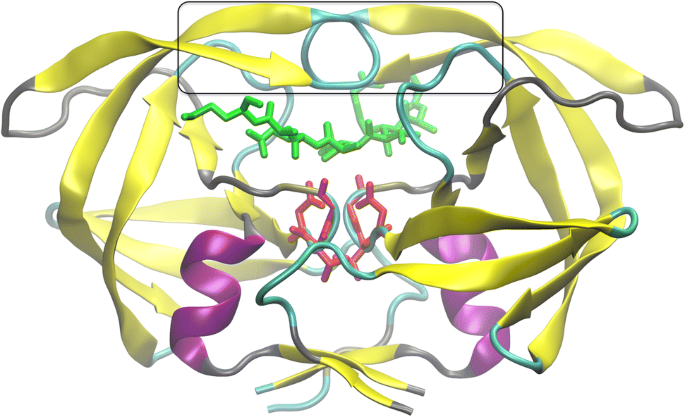
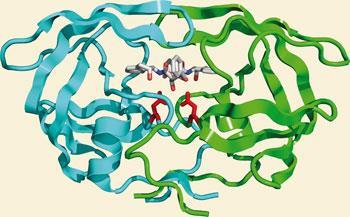
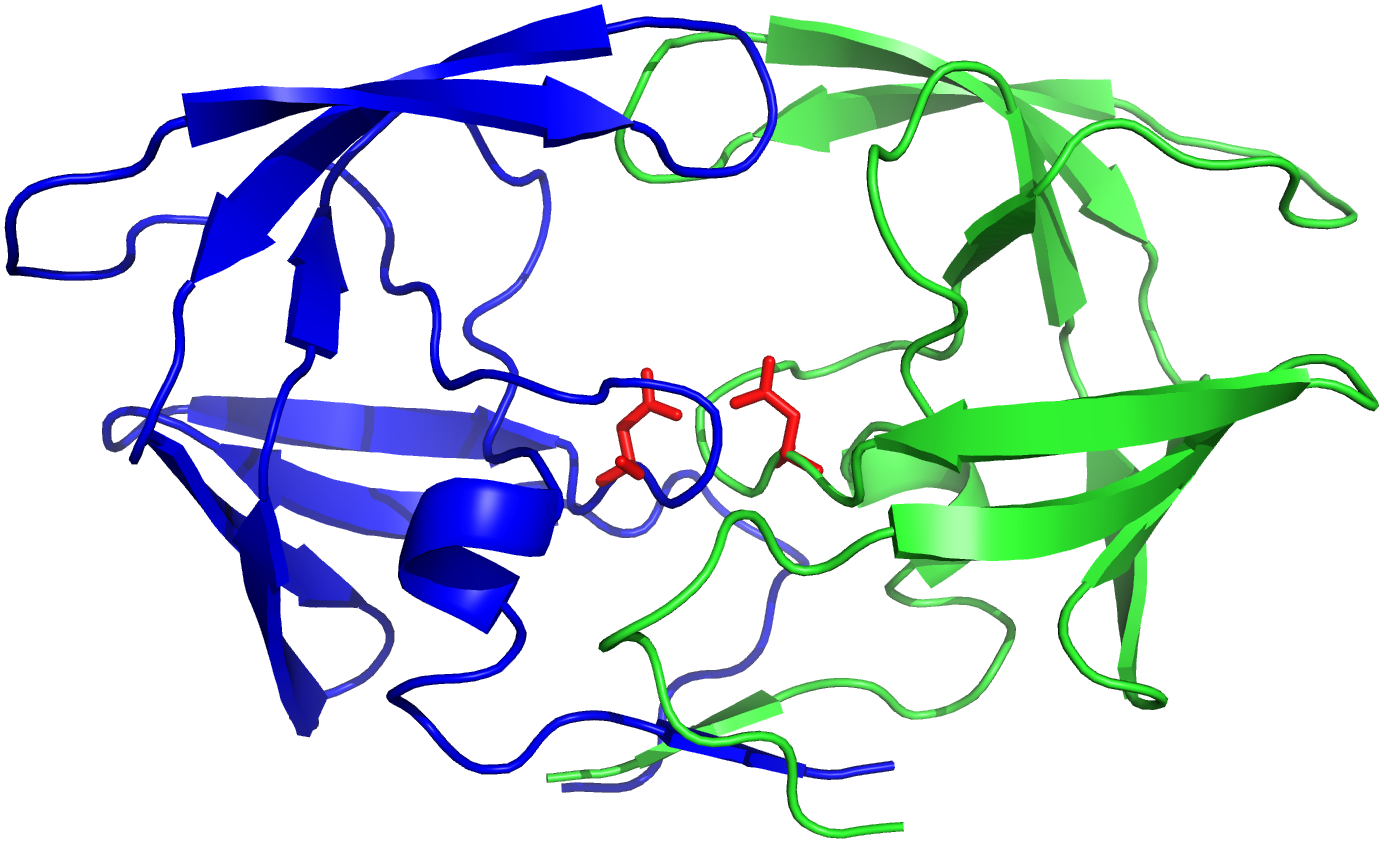
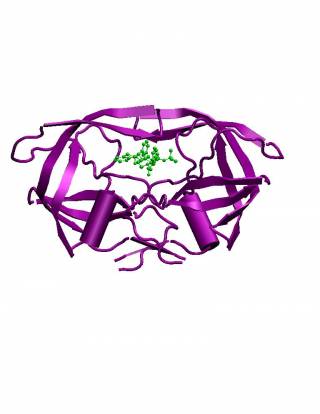
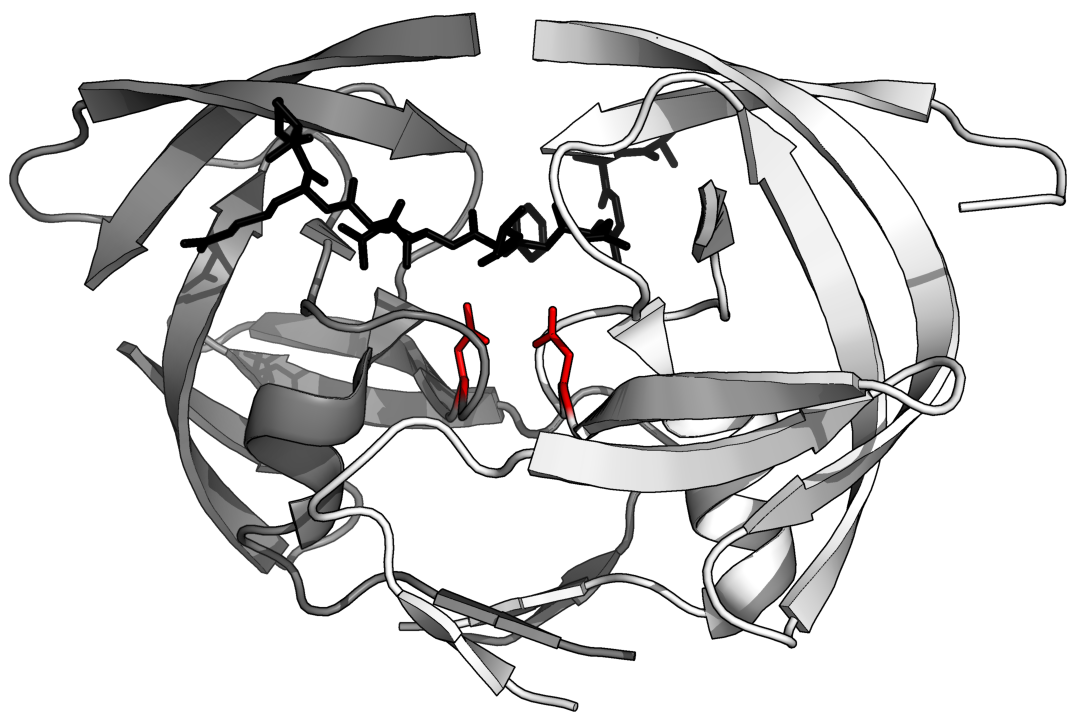
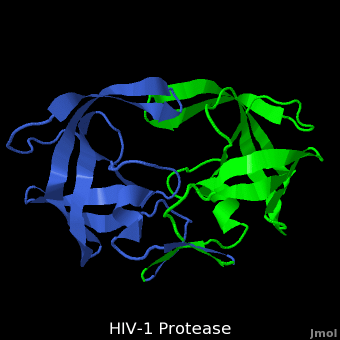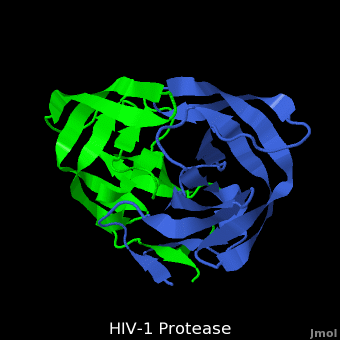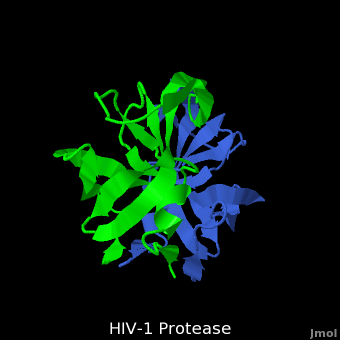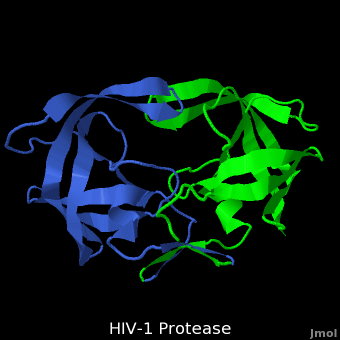
HIV-1 protease (PR) structure. Cartoon representation of PR colored by... | Download Scientific Diagram

Inhibition of autoprocessing of natural variants and multidrug resistant mutant precursors of HIV-1 protease by clinical inhibitors | PNAS
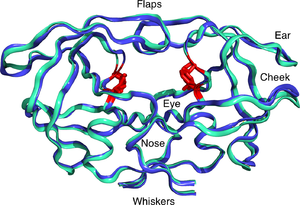
HIV Protease Hinge Region Insertions at Codon 38 Affect Enzyme Kinetics, Conformational Stability and Dynamics | The Protein Journal

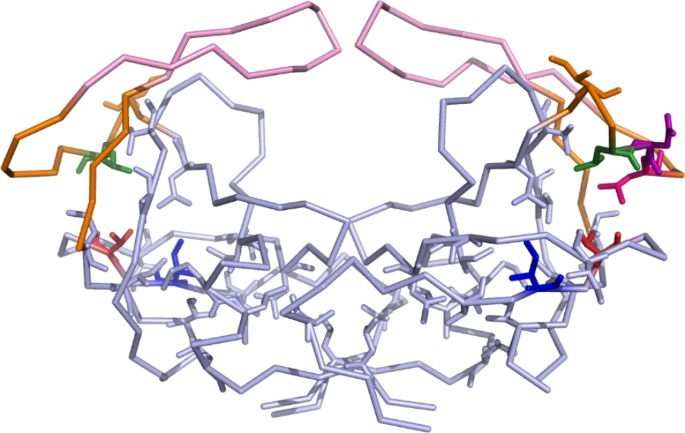
HIV-1 Protease Dimerization Dynamics Reveals a Transient Druggable Binding Pocket at the Interface | Scientific Reports

Visualizing Tetrahedral Oxyanion Bound in HIV-1 Protease Using Neutrons: Implications for the Catalytic Mechanism and Drug Design | ACS Omega
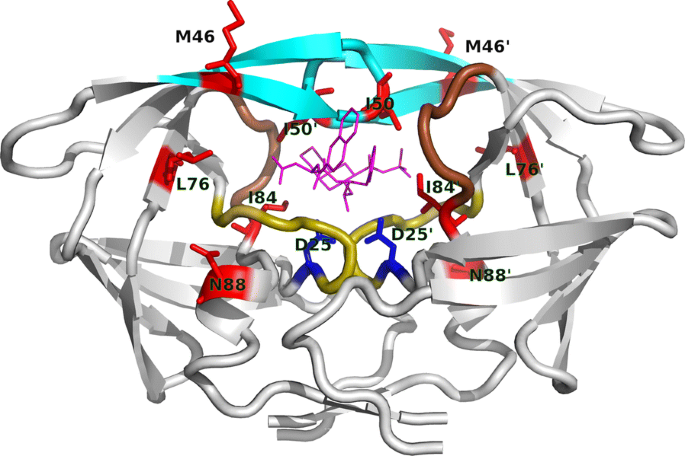
Non-active site mutants of HIV-1 protease influence resistance and sensitisation towards protease inhibitors | Retrovirology | Full Text

Drug Resistance Conferred by Mutations Outside the Active Site through Alterations in the Dynamic and Structural Ensemble of HIV-1 Protease | Journal of the American Chemical Society

Structure of HIV-1 protease in complex with potent inhibitor KNI-272 determined by high-resolution X-ray and neutron crystallography | PNAS

Characterizing early drug resistance-related events using geometric ensembles from HIV protease dynamics | Scientific Reports
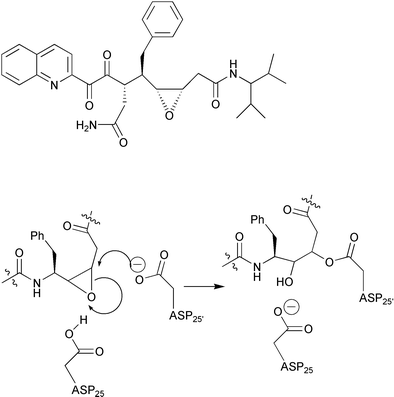
HIV-1 protease: mechanism and drug discovery - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B208248A
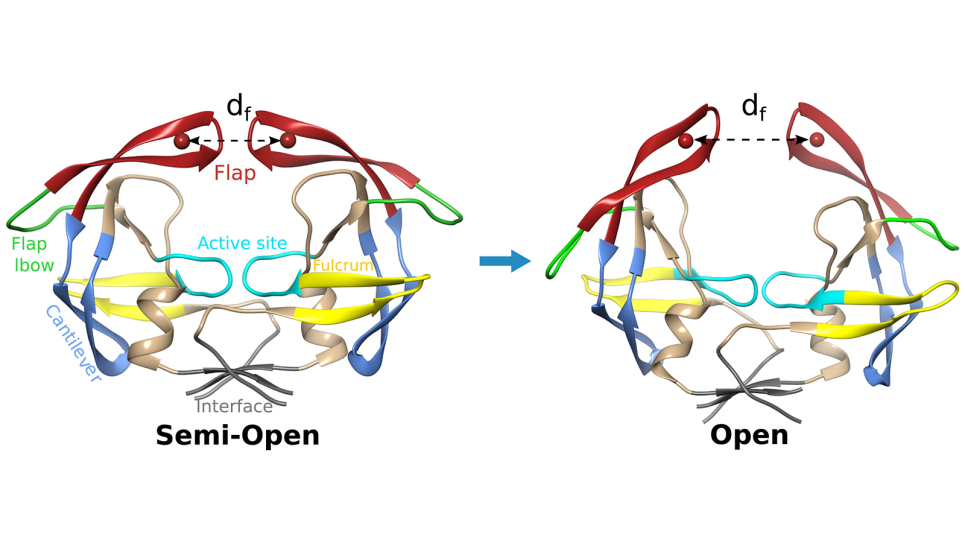
Scientists use Argonne supercomputer to detail HIV protein mechanism crucial for drug development | Argonne Leadership Computing Facility